Some of the men who built Stonehenge have been found. Their grave, which dates to the beginning of the Bronze Age, about 2,300 BC, was found at Boscombe Down near to Stonehenge.
Many of the stones at Stonehenge were brought from Wales at about this time. Chemical tests on the teeth of the men, called the Boscombe Bowmen, have shown that they were almost certainly born in Wales.
In medieval times, people believed that the stones could only have been brought to Stonehenge by Merlin the Wizard. Now archaeologists have found the mortal remains of one of the families who were almost certainly involved in this monumental task.
This section gives details, including the results of tests carried out to find out as much information about the Boscombe Bowmen as possible.
The discovery
The grave of the Bowmen was discovered during the digging of a trench for a new water pipe in May 2003. When archaeologist Colin Kirby saw human bone and pottery dating to the Bronze Age pottery, he realised immediately that a grave had been disturbed.
It was soon clear that there was something unusual about the grave. There was not just one skeleton, there were also bones from several other people. It was also clear that while the water trench had cut down one side of the grave, a trench for an electricity cable had also cut across part of the grave. What had survived the electricity and water trenches was quite unexpected.
The discovery of the Boscombe Bowmen follows hard on the heels of the astonishing story of the Amesbury Archer. The Archer was given the richest burial of his age. He was the earliest metalworker in Britain, and a man who came from central Europe. Like the Boscombe Bowmen, he lived at the beginning of the Bronze Age, at a time when great building works were going on at Stonehenge.
Wessex Archaeology is a practice of archaeologists based near Salisbury, Wiltshire, England. It provides a wide variety of archaeological services for clients including housing developers and heritage organisations. The services include excavation, evaluation, assessments of existing records, marine archaeology and building surveys. It is also a registered charity with a remit to educate the public in archaeology.
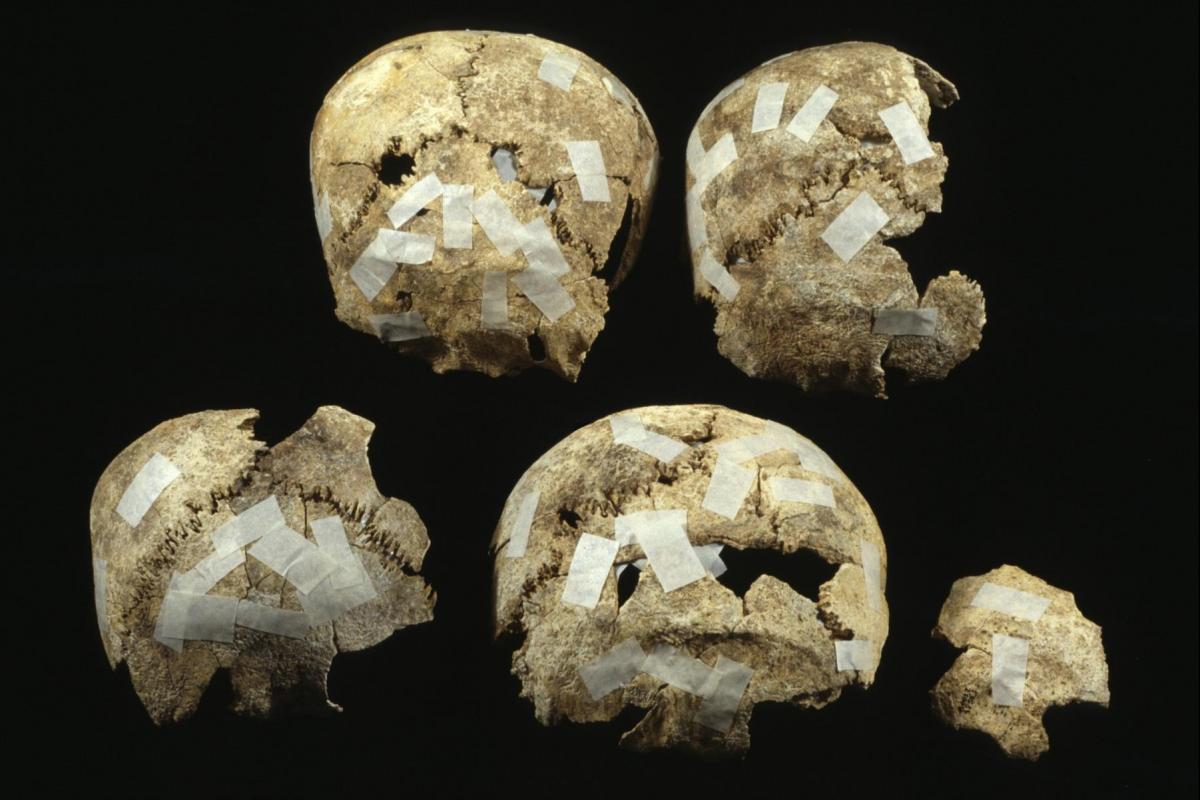

A mass grave
Most graves that date to the early in the Bronze Age (or the Beaker period) in southern England usually contain one, sometimes two, skeletons.
The Boscombe Down grave was different. A normal sized grave contained the remains of seven individuals. Three adult males, a teenage male and three children. The burial rite was unusual.
A man who had died between the ages of about 30-45 had been buried on his left side with his legs tucked up and with head to the north. His left thighbone had been broken so badly that when his leg healed it may have been shorter. But he survived, limping along. Buried close to his head were the remains of the three children.
One child, aged between about 2-4, had been cremated but there was barely a handful of their bones in the grave. This was the only cremation burial. Little survived of the remains of a child aged between about 5-6, perhaps because this inhumation burial had been disturbed. As the burial of the third child, aged between about 6-7, lay at a higher level, it would seem to have been inserted into the grave at a later date.
In contrast the remains of the teenager and the two men had clearly been rearranged. The teenager was probably a male and died between the ages of about 15-18. The two men had died between the ages of about 25-30.
Their remains had been placed around the corpse of the 30-40 year old. Some bones had been placed below his body, others on top, with similar bones being grouped together. The bones were mainly robust long bones, and parts of skulls. There were few smaller bones. The skulls were placed towards the feet of the articulated burial.
The surfaces of the loose bones were more worn. It seems likely that this was not their first resting place. The skulls of the men and the teenager were very similar in shape. All of them had distinctive small bones (called Wormian bones) that had grown between the individual parts of the skull. It seems highly likely that the males were all related.
This is a typical Stone Age rite of collective burial. But this collective burial was in a single grave, something typical of the metal ages.


The finds
Matching the seven individuals were eight pots. This is the greatest number of people from a single Beaker grave in Britain and it is also the greatest number of Beaker pots from one grave. This is not likely to be a coincidence.
Four of the pots were placed near the head of the articulated burial. Above his face were two pots. Behind his head, and close to the burials of the children, was a matching pair of pots. The smaller pot of the two had been placed inside the larger one.
Parts of one pot lay by the man’s feet but the remainder of it had already been disturbed. The remaining four vessels were all retrieved from the spoil heap. It can only be said that it is likely that they had been placed behind the man.
Seven of the eight pots are decorated all over, six with cord, one with plaited cord. The eighth pot is fragmentary but it too may have been decorated all over this time in horizontal zones with a mixture of cord decoration and combed decoration.
Such a large number of pots decorated in this way is a very unusual find. Plaited cord is an extremely rare form of decoration on Beakers in Britain. It is much more common in continental Europe. One of the very few British finds is in the nearby grave of the Amesbury Archer.
The similarity between the pots in these two graves suggests that they are about the same date. The radiocarbon dates for the Bowmen are not yet available, but on the basis of the dates from the Archer, the Bowmen’s grave is also likely to date to between 2,400-2,200 BC.
The other finds include five barbed and tanged arrowheads - giving the name the Boscombe Bowmen – some other flint tools; scrapers and flakes, a boar’s tusk and a toggle.
In continental Europe tusks are often found in the same grave as stones used for metalworking, like the one found in the grave of the Amesbury Archer. Sometimes the tusks and stones have been found next to each other together. Are the boar’s tusks metalworking tools?
Only one other bone toggle has been found in Britain. It comes from a later, and rich, Bronze Age burial at Barnack, Cambridgeshire. Most toggles have been found in continental Europe. These finds usually have a central perforation rather than a loop. They have mainly been thought of as decorative pendants but they may have fastened clothing or a hair ornament.
Both the decoration of the plaited cord beaker and the bone toggle point towards continental Europe, from where the Amesbury Archer came. Some of the objects buried with him also came from there. But the Boscombe Bowmen came from Britain.
Men from the west
Like the Amesbury Archer, the enamel on the Bowmen’s teeth has provided the clue as to where they grew up. As different teeth form at different times in childhood every tooth preserves a chemical fingerprint of the local environment at that time.
Pre-molars form after the milk teeth have dropped out, between the age of 3 and 6. Third molars are the last teeth to erupt and form between 9-13.
When the tooth enamel is fixed, the oxygen and strontium isotopes in it are locked into the chemical fingerprint. For pre-molars, this is in early childhood. For the 3rd molars, it is what may be early adolescence.
Although the oxygen isotopes of the Amesbury Archer showed that he came from continental Europe. Other Beaker age people buried near to Stonehenge have been shown to be local. These are burials Stonehenge itself, from Normanton Down, and the Amesbury Archer’s Companion.
Analysis by the British Geological Survey showed that the Boscombe Bowmen’s teeth have a particularly high proportion of the strontium isotope, which shows that grew up in a place with a very high radiogenic, or radioactive background. The radioactivity from the underlying rocks passes into the soils and from there into the food chain.
Such high strontium levels can only be matched in a few places in Britain. In Cornwall, the Isle of Man, the north-west of England, parts of the Scottish Highlands, and Wales.
The oxygen isotopes in our teeth come from drinking water. The character of the isotopes varies according to a number of factors including distance from the sea, height above sea level etc. Because of this, oxygen isotopes are good indicators of the climate a person was living in when the enamel on their teeth formed.
The effect of climate change on oxygen isotopes has to be considered but the changes since the last Ice Age have been too small to make a significant difference.
The Bowmen’s oxygen isotopes are not compatible with the environments of Cornwall and the Isle of Man as they are too warm. Most of Scotland is too cool. The only areas that agree with the strontium levels are the Lake District and north Wales and south-west Wales.
But not only it is possible to show that the Bowmen were in one of these places when they were very young, it can be shown that they migrated. The chemical fingerprints of each Bowman have exactly the same differences between their premolars and their 3rd molar.
The men were in one place up to the age of 6 and in another up to the age of 13. The second place may also have been in Wales. It was not in Wessex. The chemical fingerprint of both of these places is different to that of the chalk geology of Wessex.
Either the Bowmen all moved at the same time over a number of years, or their society regularly moved children between the age of 6 and 13 to live in a different place. They moved nearer to Stonehenge later in their life.
Further tests on the teeth of the children buried with the Bowmen are in progress, but this is already a remarkable picture of prehistoric migration. It is a picture that is entirely consistent with the close similarities in the shape of the men’s skulls that suggests that they are all related.
The bluestones of Stonehenge
The stone circles of Stonehenge are built from two main types of rock. The massive sarsens that are a sandstone, and a variety of smaller igneous rocks known as the bluestones. It has been known since the 16th century that the sarsen stones came from near to Marlborough, 30 km north of Stonehenge.
It was only 350 years later that the source of the bluestones was pinpointed. The three main types of bluestone come from the Preseli Hills in north Pembrokeshire. On Carn Menyn some of the stones could simply have been collected from the surface.
It has been argued that the stones were transported from Wales by glaciers. However, comprehensive geological studies have shown that there is no evidence for a glaciation in Wessex that could have transported these rocks and left no other trace.
When the stones were transported to Stonehenge and added to the temple that already stood there is less easy to establish. The stones helped to transform the layout of the monument so that it was aligned on the sunrise on the longest day of the year; and sunset on the shortest day.
The bluestone settings at Stonehenge are thought to have been re-arranged at least four times within a period about 400 years between 2,400 and 2,000 BC. There may have been plans for a fifth arrangement that was not completed.
The date at which the bluestones first arrived at Stonehenge is not known. It can only be said that the first bluestone setting (Phase 3i: the Q and R holes) is earlier than the completion of the setting of great sarsen stones, which is radiocarbon dated to 2,440-2,100. Pieces of beaker pottery found in the backfill of one of the stoneholes of this first bluestone setting show that Beaker pottery was in use when that stone was removed.
It is not known whether all the bluestones arrived at the same time. Richard Atkinson, who with Stuart Piggott, was the most recent excavator at Stonehenge, favoured this option. But Ros Cleal, Karen Walker and Becky Montague who published those excavations, preferred to see the bluestones as having arrived in two major episodes. The second episode being after the sarsen settings had been erected and when the Bluestone Circle and Bluestone Oval were built. The Bluestone Circle is radiocarbon dated 2,280-2,030.
The Preseli Hills, source of the Stonehenge bluestones, lie within one of the highly radiogenic areas in which the Boscombe Bowmen were born.
This may be a coincidence, but it seems highly probable that the men and their families were involved in the movement or migrations associated with or established by the movement of the bluestones.

Why were the bluestones brought from Wales?
Centuries of speculation have not yielded a definitive conclusion. But some things now look more likely.
One possibility is that a stone circle was brought from Wales. One of the very few places that an oval stone setting similar to the Stonehenge Bluestone Oval can be found is high in the Preseli Hills at Bedd Arthur. A similar oval setting is known at The Churchyard on Skomer Island, just off the south-west coast Pembrokeshire.
Even if the similarity between Bedd Arthur and Stonehenge is not a coincidence, there are many, many, more bluestones at Stonehenge than in the stones of these other oval settings. The transport of a single monument to Stonehenge does not seem to provide the complete solution for the Stonehenge bluestones.
Earlier writers have looked to the connection between metalworkers prospecting for ores. David Clarke wrote of Beaker period warriors ‘who may have had the Prescelly stones brought along the copper/gold route to Wiltshire and to Stonehenge’. Clarke was thinking of people who used a later style of beaker pottery (his Wessex/Middle Rhine type). These people were thought to be invaders.
In south-west Ireland there is evidence for copper mining on Ross Island in County Kerry that started between 2,400-2,200 BC. Beaker pottery has been found in the mines. Both metallurgy and Beaker pottery were introduced to Ireland at this time. It seems likely that the same group of people introduced both.
In Britain the earliest metalworker, the Amesbury Archer, an incomer from central Europe, lived between 2,400-2,200 BC. All these dates, from Stonehenge, from Ross Island, and the Amesbury Archer, lie in the earlier part of second half of the 3rd millennium BC. At Stonehenge, Beaker pottery is associated with all the major stone settings.
In trying to explain why the bluestones were moved, Richard Atkinson suggested that for travellers between Wales and Ireland, Carn Menyn was a landmark from land and sea; and a sacred place.
‘I believe, therefore, that the awe-inspiring character of Prescelly Mountain is alone sufficient to account for the special significance of the rocks which crop out along its crest.’
The legend related by Geoffrey of Monmouth in about 1136 in his Historia Regium Britanniae that Merlin the Wizard brought the stones to Stonehenge by transporting a stone circle from Ireland retains elements of the original story.
The legend tells of travel, of transformation...and of magic
Today we can tell that the stones of Stonehenge were brought from the west to transform an existing temple.
The families of the Boscombe Bowmen brought the bluestones with their own hands.
Men like the Amesbury Archer travelled from afar to Stonehenge. They brought the new and magical skill of metalworking in their hands and their head.
Then, metalworkers could change rock to metal, a change of substance and colour, by the magic of fire.
Now, it seems more and more likely that the builders of Stonehenge transformed a timber monument to one in stone by bringing the stones back from the route of their voyages to an island of metals.
An island that lay where the sun set in a blaze of copper gold.
The building of Stonehenge
Stonehenge had been a temple for a thousand years before the stones were erected. It is likely that timber settings stood before the stones were brought to the site.
Following the phasing proposed in the definitive excavation report on the 20th century excavations at Stonehenge by Ros Cleal, Karen Walker and Becky Montague, the first stone setting at Stonehenge was Phase 3i.

Phase 3i
This was a double arc of bluestones in what are known as the Q and R holes. Richard Atkinson had previously thought that they had formed concentric circles but there is only evidence for an arc, perhaps with other settings where the arc was not continued. Apart from the bluestones there is a much larger sandstone from Wales at Stonehenge. Known as the Altar Stone, this stone probably came from near to Milford Haven on the coast to the south of the Preseli Hills, and it may have stood as a large standing stone in this phase. When this setting was built is not known for certain, nor when it was dismantled though a sherd of comb decorated beaker pottery was found in the backfilling of one of the stone holes (Q5).
Phase 3ii
The next arrangement of stones (Phase 3ii) was the great Sarsen Circle with a horseshoe shaped setting of even larger stones, the Trilithons, inside it. These settings, which form the classic image of Stonehenge, are thought to have been built at the same time and despite their names, both the Sarsen Circle and the Trilithons have trilithons, or horizontal stones, on top of them. The accepted radiocarbon dates for this phase are 2440-2100 BC.
Phase 3iii
It is quite possible, but not proven, that the bluestones that stood in the Phase 3i Q and R holes were taken down at this time and rearranged into a new setting that stood within the Phase 3ii sarsen settings. This phase of bluestones, known as phase 3iii, is very poorly documented and its arrangement is not known. It is thought, however, that it may well have incorporated the bluestone trilithons, stones from which were reused in the later Phase 3iv settings.
Phase 3iv
The bluestones were rearranged again in Phase 3iv. A circle of single bluestones, the Bluestone Circle, was set up between the Sarsen Circle and the Trilithons. It is also thought that an oval setting of bluestones was built within the horseshoe shape of the Sarsen Trilithons. It seems likely that the Altar Stone was moved to stand within this Bluestone Oval. The bluestones trilithons that are thought to have been used in Phase 3iii are found reused in both the Bluestone Circle and the Bluestone Oval. It is thought that further bluestones could have been introduced at this time. The Avenue that extends between the River Avon and Stonehenge may have been have been built at this time. The Bluestone Circle is dated to 2280-1930 BC.
Phase 3v
Phase 3v saw the arrangement of the bluestones that stands today. Part of the northern arc of the Phase 3iv Bluestone Circle was removed, creating a horseshoe shaped setting known as the Bluestone Horseshoe. This echoes the shape of the sarsen Trilithons. The Bluestone Horseshoe is dated to 2270-1930 BC.
Phase 3vi
The final phase of stone settings, Phase 3vi, is represented by the Y and Z holes. These were dug in concentric circles outside the Sarsen Circle but they never held stones. It is possible that it was intended to reposition the existing bluestones in the Y and Z holes or to bring in new bluestones, but this did not take place. The e Y and Z holes date to early in the 2nd millennium.
Strontium isotope analysis
The strontium isotope composition of tooth enamel can provide information about where an individual spent their child hood. Strontium isotopes provide a fingerprint for different rock types and, as the distribution of rocks is well mapped in Britain and around the world, the geology provides the key to geographic location.
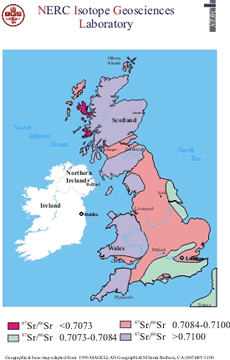
87Sr is formed by radioactive decay of 87Rb and it accumulates slowly through geological time. The 87Sr is normally expressed as a ratio against the stable 86Sr isotope as the 87Sr/86Sr ratio. In rocks that are old (>100 million years) and particularly if they also contain high levels of rubidium, the 87Sr/86Sr isotope ratio is high. This is typical of old granite and gneiss terrains. In rocks that are geologically young (<100 million year) and have low rubidium content the 87Sr/86Sr ratio is low. Rocks of this type tend to be recently erupted basalts. These values are transmitted, through the weathering of rocks to form soil, into the biosphere via plants and in drinking water. The range of 87Sr/86Sr ratios may seem very small, a typically low value would be 0.704 whereas 0.720 would be considered high, but such differences are easily measured to high levels of precision. The Bowmen’s teeth have a very high 87Sr/86Sr ratio.
Ideally, as with the Boscombe Bowmen, strontium isotope analysis is used in conjunction with other lines of evidence such as oxygen isotope analysis to constrain possible areas where an individual could have spent their childhood and/or rule out areas where the tooth data does not match environmental values.
Dr Jane Evans - British Geological Survey



